Thousands of Australian families coming to terms with Vaginal Mesh scandal
It is now emerging that thousands of women in Ireland and worldwide have suffered traumatic complications as a result of vaginal mesh surgery.
A number of women recently won their class-action lawsuit in Australia against Ethicon, a subsidiary of Johnson & Johnson. This was one of Australia’s largest ever product liability class actions.
The Australian Federal Court found that Ethicon had been negligent and had engaged in misleading conduct and hid the risks associated with the implants from patients.
What are Vaginal Mesh implants?
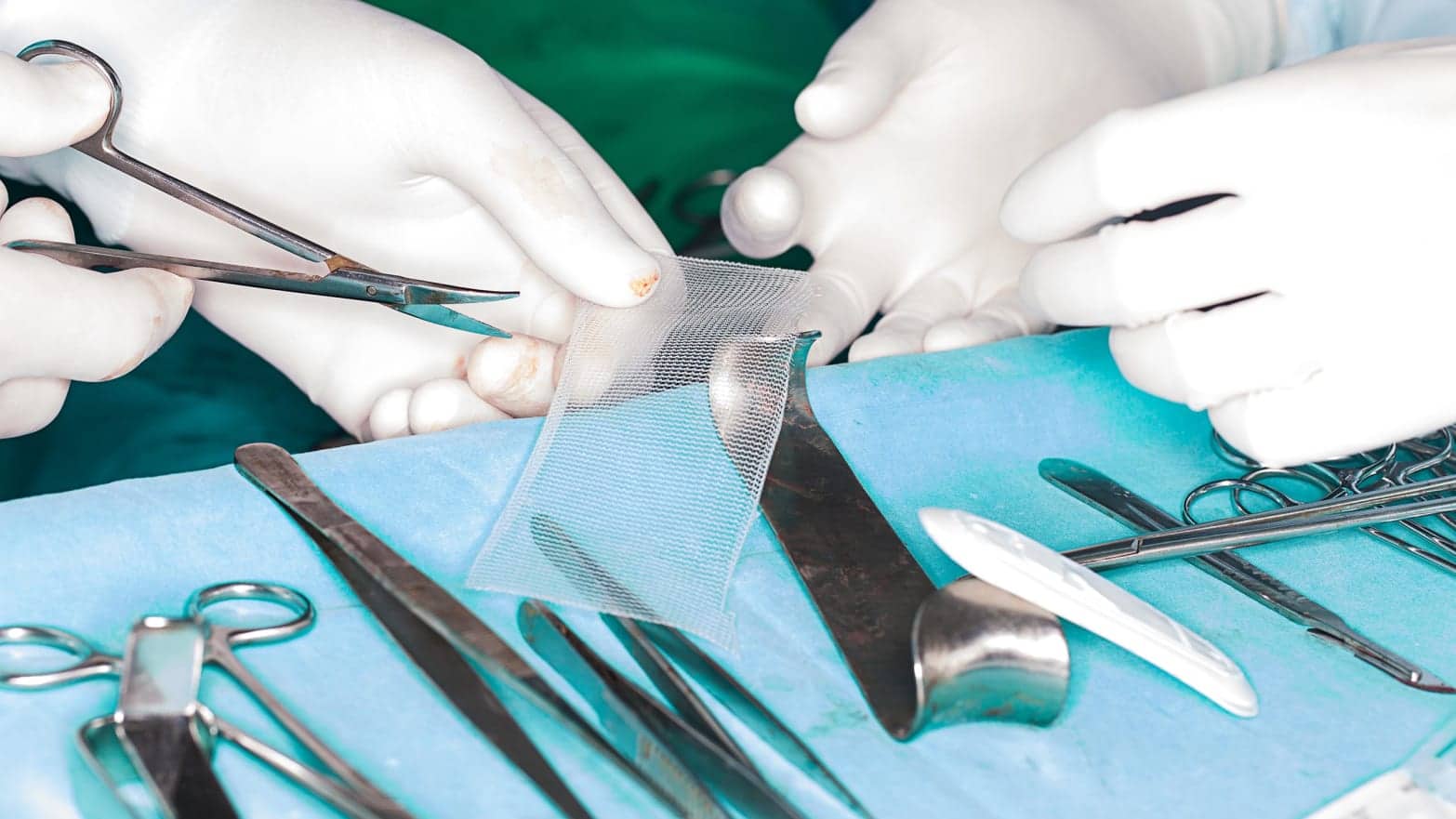
Vaginal Mesh implants are non-absorbable implants, made of synthetic netting that are implanted in a surgical procedure to treat a number of conditions, most commonly pelvic organ prolapse and stress urinary incontinence.
What are the possible complications?
The Australian Federal Court listed the adverse events of the implants:
- Infection
- Erosion of the mesh into the vaginal canal resulting in infection which may be difficult to treat and cause offensive vaginal discharge and pain
- Erosion of the mesh into surrounding organs such as the bladder, urethra or rectum which may cause pain and damage those organs
- Damage to nerves in the scar tissue surrounding the implant or elsewhere;
- Chronic pain, which may be severe
- Painful sexual intercourse, which may be severe and may become chronic or inability to have sexual intercourse
- Leg weakness
- Recurrent incontinence
- Difficulty voiding and
- Vaginal discharge.
The claimants in the Australian Class-Action lawsuit described their pain as:
“of such an intensity that she could not speak and at times struggled to breath”;
“Excruciating”; and
“something sharp, like a blade” .
One of the claimants in the Australian case felt that the Company:
“have treated women essentially like guinea pigs, lied about it and done nothing to help.”
The Decision of the Australian Federal Court.
Judge Anna Katzman found that the information provided to patients and surgeons by the Company had:
- Omitted warnings of the complications of the implants;
- Failed to include warnings about the gravity of the risks and how they could be reduced;
- Included inaccurate and at times false representations about the devices.
Examples of Ethicon’s misleading and deceptive conduct
- The assertion that a foreign body reaction ‘may’ occur, when the Company knew that it would invariably occur, which was calculated to convey a false sense of security;
- The omission of a reference to the risk of chronic pain coupled with references to the risk of transient pain, which was likely to lead to the erroneous belief that there was no prospect of chronic pain;
- The downplaying of the risk of infection and the implication that the only risk was that implantation could promote or enhance a pre-existing infection;
- The failure to warn of the increased risk of a heightened inflammatory response in immunosuppressed patients or to advise that implantation with the devices was not advised in such cases; and
- The delay in warning that revision surgery or multiple surgical procedures might be necessary. Nor did they warn of the difficulties of removing the mesh.
Judge Katzman stated that:
“ I am satisfied that, at all material times the Ethicon devices were or have been on the market, [Ethicon] engaged in a course of conduct which was misleading or deceptive or likely to mislead or deceive a not insignificant number of patients and surgeons as to both their safety and efficacy.”
Negligence:
The Australian Federal Court held that Johnson & Johnson and Ethicon breached their duty of care to the claimants. The Court stated that the risks were known not to be insignificant and on Ethicon’s own admission, they knew serious harm could ensue.
Compensation:
The level of compensation to be received by the Australian claimants will be decided on by the Court in February 2020.
It remains to be seen if Johnson & Johnson will appeal the decision.
The response of the Irish State to the Scandal
It is not clear that the scandal is receiving the attention it merits from the various state bodies who ought to take responsibility, including the Health Service Executive, the Department of Health and the Health Products Regulatory Authority.
The Chief Medical Officer recommended in July 2018 that a pause be put in place on the use of procedures involving synthetic mesh for treatment of stress urinary incontinence and pelvic organ prolapse.
The Health Products Regulatory Authority has stated that it is conducting a review of the regulatory aspects of these products as a matter of priority.
On 26th June 2018 the HSE circulated a learning notice to Hospitals concerning the use of Mesh devices in Uro-Gynaecological Procedures, noting the “controversy about the use of mesh devices” in many jurisdictions.
The Notice did acknowledge that “a significant cause of patient dissatisfaction is the difficulty in accessing services should serious complications develop. Such patients have a firmly held conviction that their needs are not being addressed to their satisfaction in Ireland.”
By way of addressing this, the HSE has convened a Response Group to address and propose remedies to these issues.
However, families report that there are few practical supports and services for those affected by the vaginal mesh implants.
How can we help?
Our medical negligence department has experience in assisting families who have been affected by defective and harmful medical products. If you or a member of your family have been affected by vaginal mesh implants, we will be happy to meet with you to explore the legal options available and to assist you in arriving at the truth of what happened.
For further advice or if you wish to discuss any other legal area please contact reception@lynchsolicitors.ie or telephone 052-6124344.
The material contained in this blog is provided for general information purposes only and does not amount to legal or other professional advice. While every care has been taken in the preparation of the information, we advise you to seek specific advice from us about any legal decision or course of action.